In the German border area with Groningen and Drenthe, the number of hectares of land on which wind energy can...
In recent seasons, the Jupiler Pro League has been able to push many players towards major competitions in Europe. Jonathan...
Did you know that your clothes can look wonderfully soft and fresh without using fabric softener? Read on and discover...
DierAnimal also cannot participate in elections. “It is a sad day for us,” says party leader Constance Adonis. “We did...
What can you do if this happens to you? According to Tim Derink, an attorney who specializes in construction law,...
Olivier Destacht can be seen in the program Special Forces on VTM from April 22. Three years after he stopped...
PAS detectors in the District of Utrecht should be given priority when issuing the nitrogen space from the Utrecht Nitrogen...
Dries Mertens was in Napoli on Wednesday. A visit that filled Neapolitans with nostalgia for the past. When he joined...
Intel is launching a Halo point onto the world: it's a prototype of a so-called nervous system, which works more...
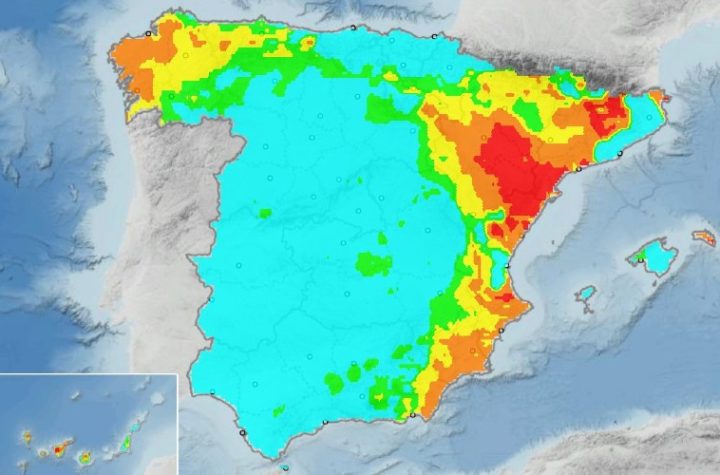
The Spanish Meteorological Service warned residents of the increasing risk of forest fires in several areas of the country. After...